|
Boyle studied the compressibility of gases in 1660. In his
experiments he observed "At a fixed temperature, the volume
of a gas is inversely proportional to the pressure exerted by
the gas." The experiment is simple: (see figure on left)
A cylinder with a piston and a gas is immersed in a bath (e.g.
water). The purpose of the bath is to have a ready heat source
to maintain the temperature of the gas constant throughout the
experiment. A mass is placed on top of the piston which results
in a pressure on the gas. The gas volume is measured and 1/V
vs P data point plotted. The mass is increased and the new 1/V
vs P data point plotted. This is continued over several larger
masses. to see what happens place the mouse cursor over the image.
The straight line implies
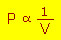
- or

Which is Boyle's law.
|

